CE FDA ISO Approved Factory Price Color Display Bluetooth USB Medical Portable Breathing Diagnostic Spirometry Machine Digital Spirometer - China Portable Spirometer, Medical Equipment | Made-in-China.com

SMART ONE Digital Spirometer - Peak Flow Meter (PEF) & FEV1 with FREE Mobile APP - by MIR | B-Arm Medical Smartone Home Use Spirometer by MIR
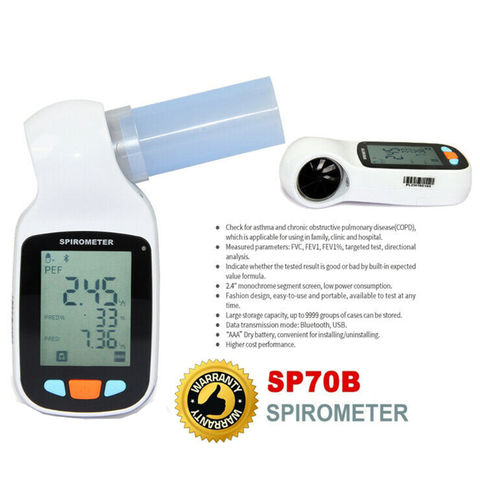
Buy Wholesale China Sinohero Handheld Medical Bt Spirometer Spirometro Portable Digital Lung Breathing Spirometer & Spirometer at USD 79 | Global Sources
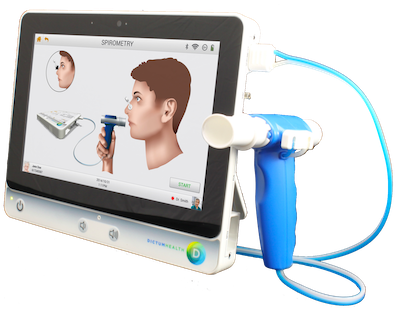
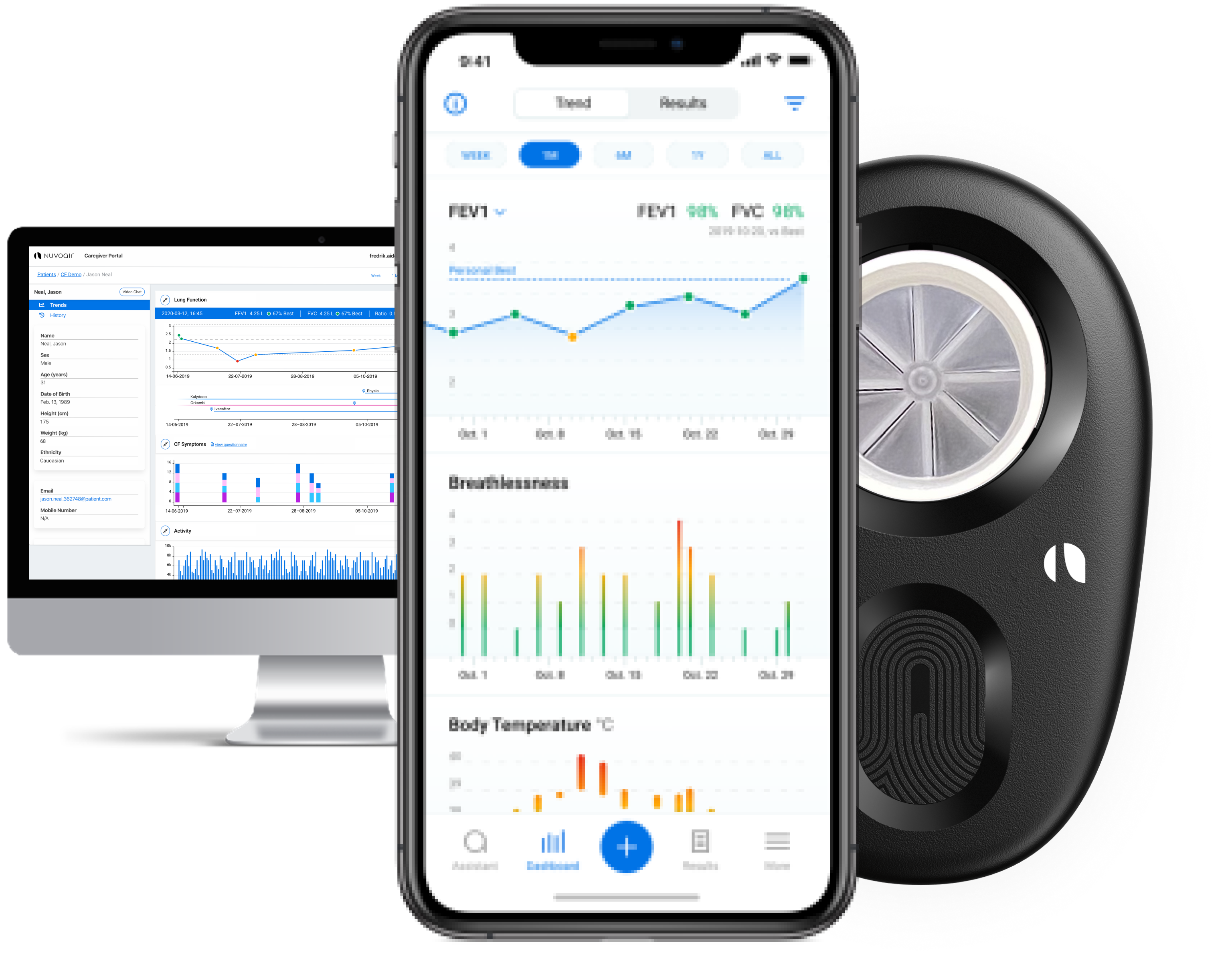
Dictum Health Receives FDA 510K Clearance for Portable Spirometry for In-Clinic Use and Remote Patient Monitoring - ATA

Aluna - Our FDA-Cleared Digital Spirometer is available for purchase at https://hubs.ly/H0pPysG0. As a member of our social community, you have early access! Now kids with Asthma or Cystic Fibrosis can measure
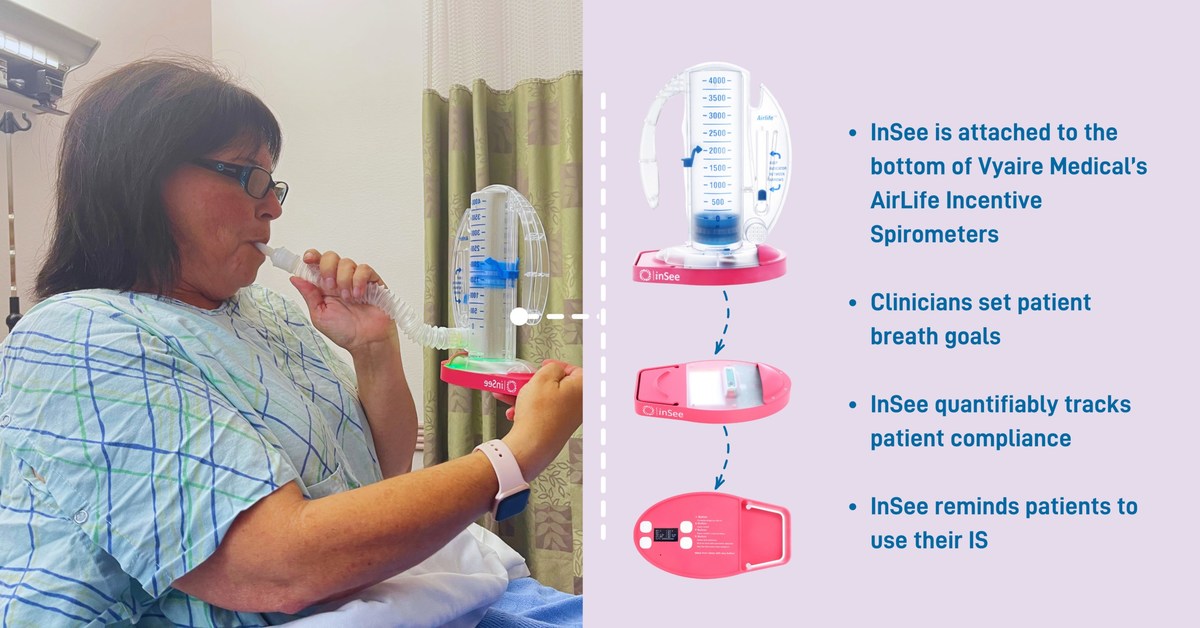
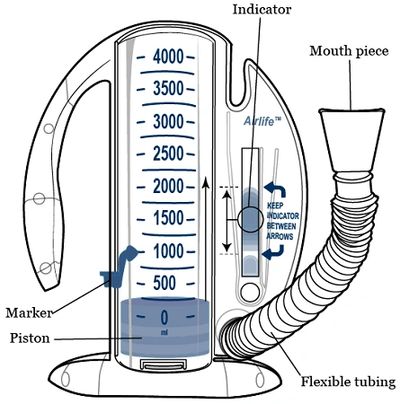
Tidal Medical Technologies receives FDA Emergency Use Authorization (EUA) for the use of InSee for tracking of incentive spirometers usage in treatment of respiratory conditions in COVID-19 patients.