Major Bleeding in Patients With Coronary or Peripheral Artery Disease Treated With Rivaroxaban Plus Aspirin - ScienceDirect

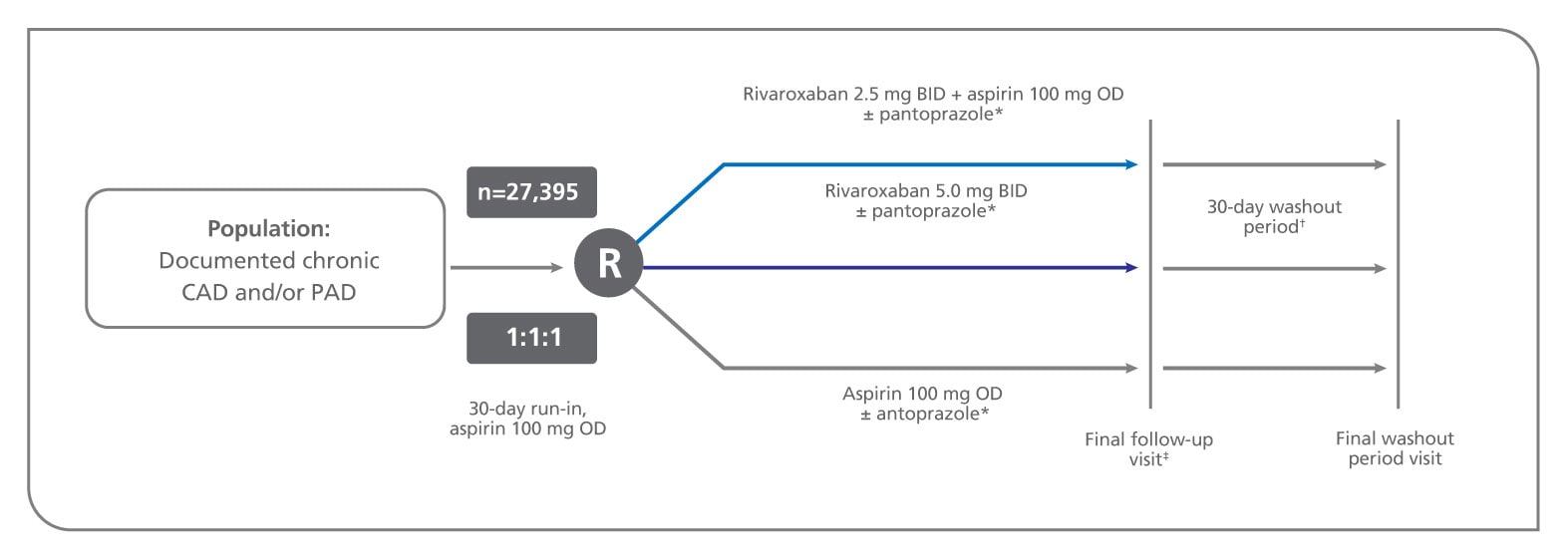
Figure 2, COMPASS Design Schematic - Clinical Review Report: Rivaroxaban ( Xarelto) - NCBI Bookshelf

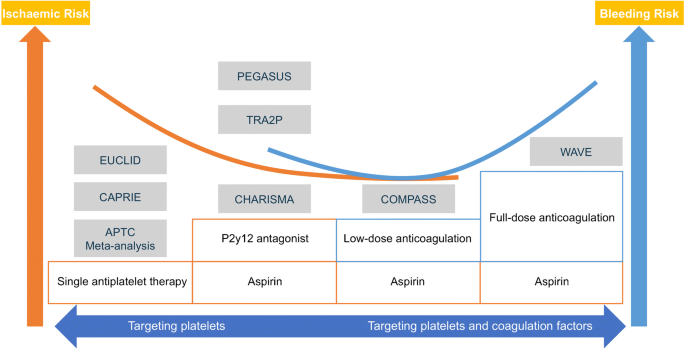
Rivaroxaban Plus Aspirin Versus Aspirin in Relation to Vascular Risk in the COMPASS Trial | Journal of the American College of Cardiology

Rivaroxaban Plus Aspirin in Obese and Overweight Patients With Vascular Disease in the COMPASS Trial - ScienceDirect

COMPASS Trial: Absolute Risk Differences Over Time for Severe Bleeding... | Download Scientific Diagram

Eligibility for Low-Dose Rivaroxaban Based on the COMPASS Trial: Insights from the Veterans Affairs Healthcare System | SpringerLink

COMPASS Trial: Incidence Rates of Different Categories of Major Bleeding | Download Scientific Diagram

Rivaroxaban Plus Aspirin Versus Aspirin in Relation to Vascular Risk in the COMPASS Trial - ScienceDirect

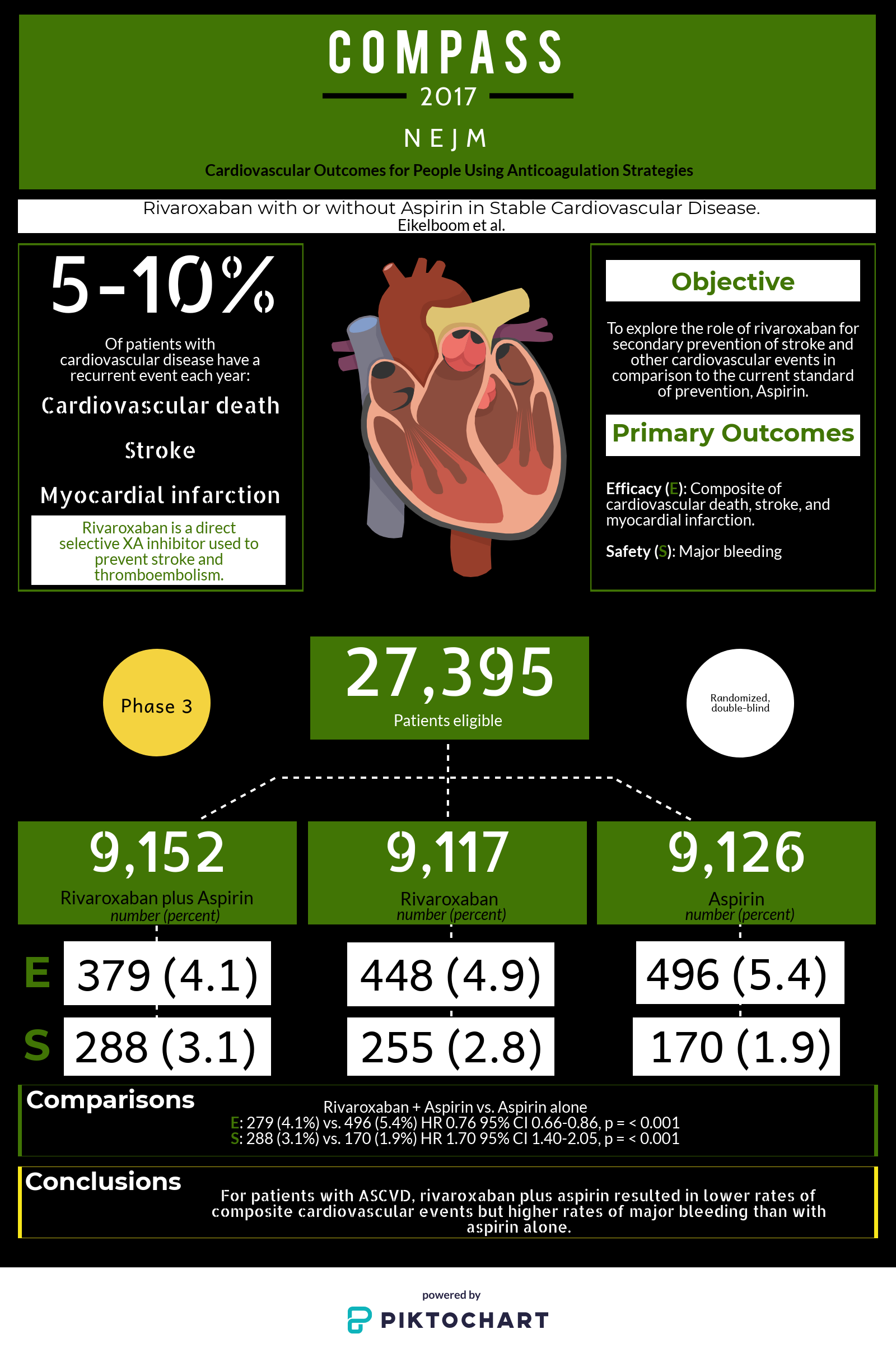
Rationale, Design and Baseline Characteristics of Participants in the Cardiovascular Outcomes for People Using Anticoagulation Strategies (COMPASS) Trial - ScienceDirect

Rivaroxaban Plus Aspirin in Patients With Vascular Disease and Renal Dysfunction: From the COMPASS Trial | Journal of the American College of Cardiology

Rivaroxaban with or without aspirin in patients with stable peripheral or carotid artery disease: an international, randomised, double-blind, placebo-controlled trial - The Lancet