A Thermodynamic Model for Multivalency in 14-3-3 Protein–Protein Interactions | Journal of the American Chemical Society

14-3-3σ binds phosphorylated COP1. (A) The consensus 14-3-3 binding... | Download Scientific Diagram
The dynamic and stress-adaptive signaling hub of 14-3-3: emerging mechanisms of regulation and context-dependent protein–protein interactions | Oncogene

Interaction of 14-3-3 proteins with the Estrogen Receptor Alpha F domain provides a drug target interface | PNAS
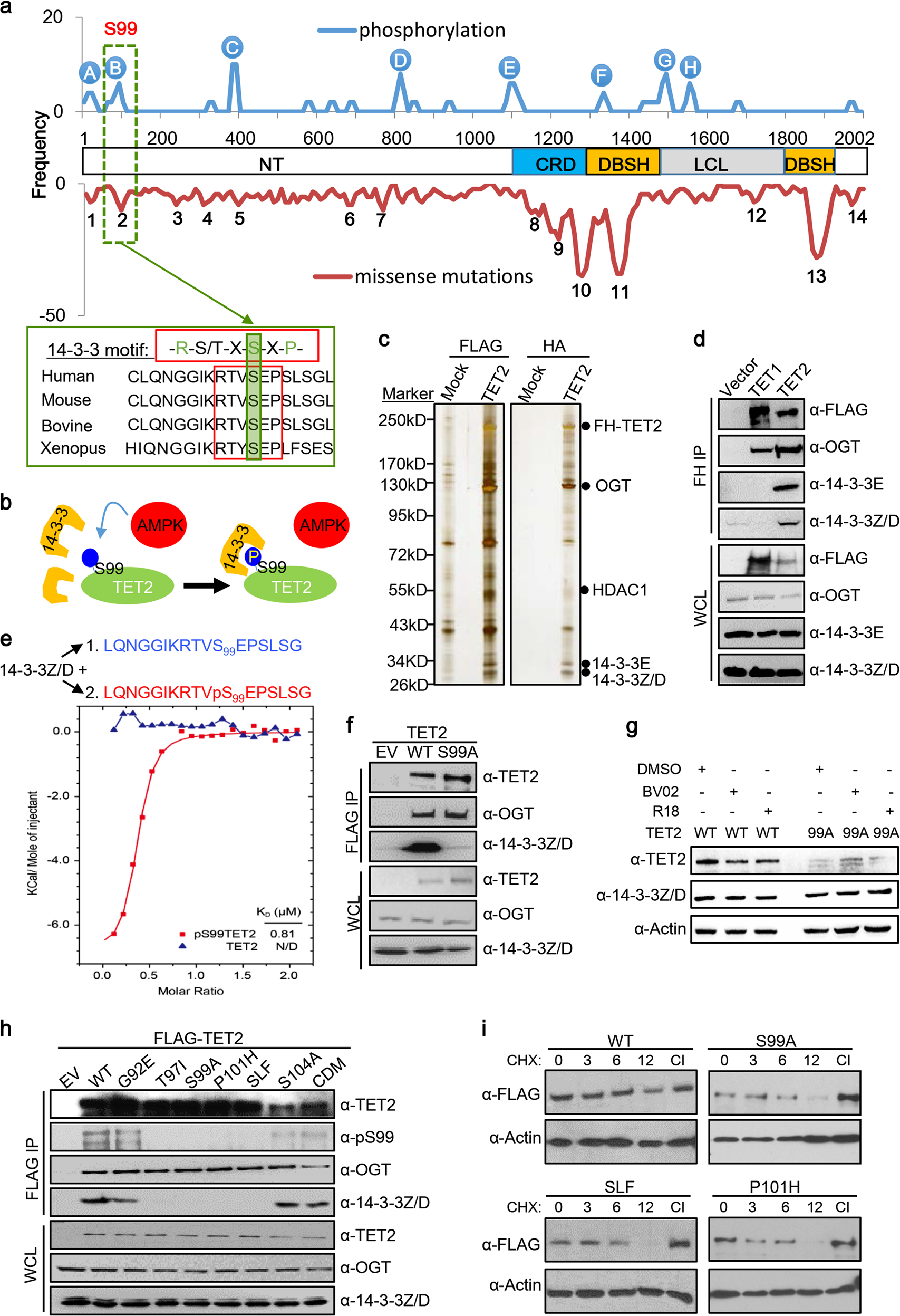
TET2 stabilization by 14-3-3 binding to the phosphorylated Serine 99 is deregulated by mutations in cancer | Cell Research

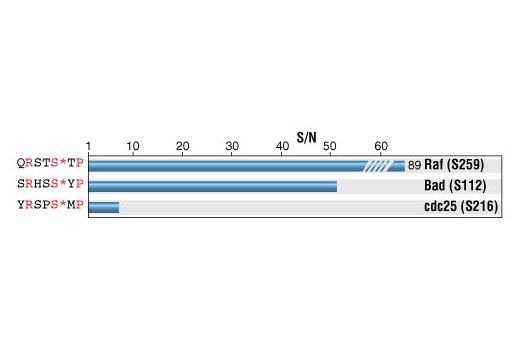
A Robust Protocol to Map Binding Sites of the 14-3-3 Interactome: Cdc25C Requires Phosphorylation of Both S216 and S263 to bind 14-3-3 - ScienceDirect

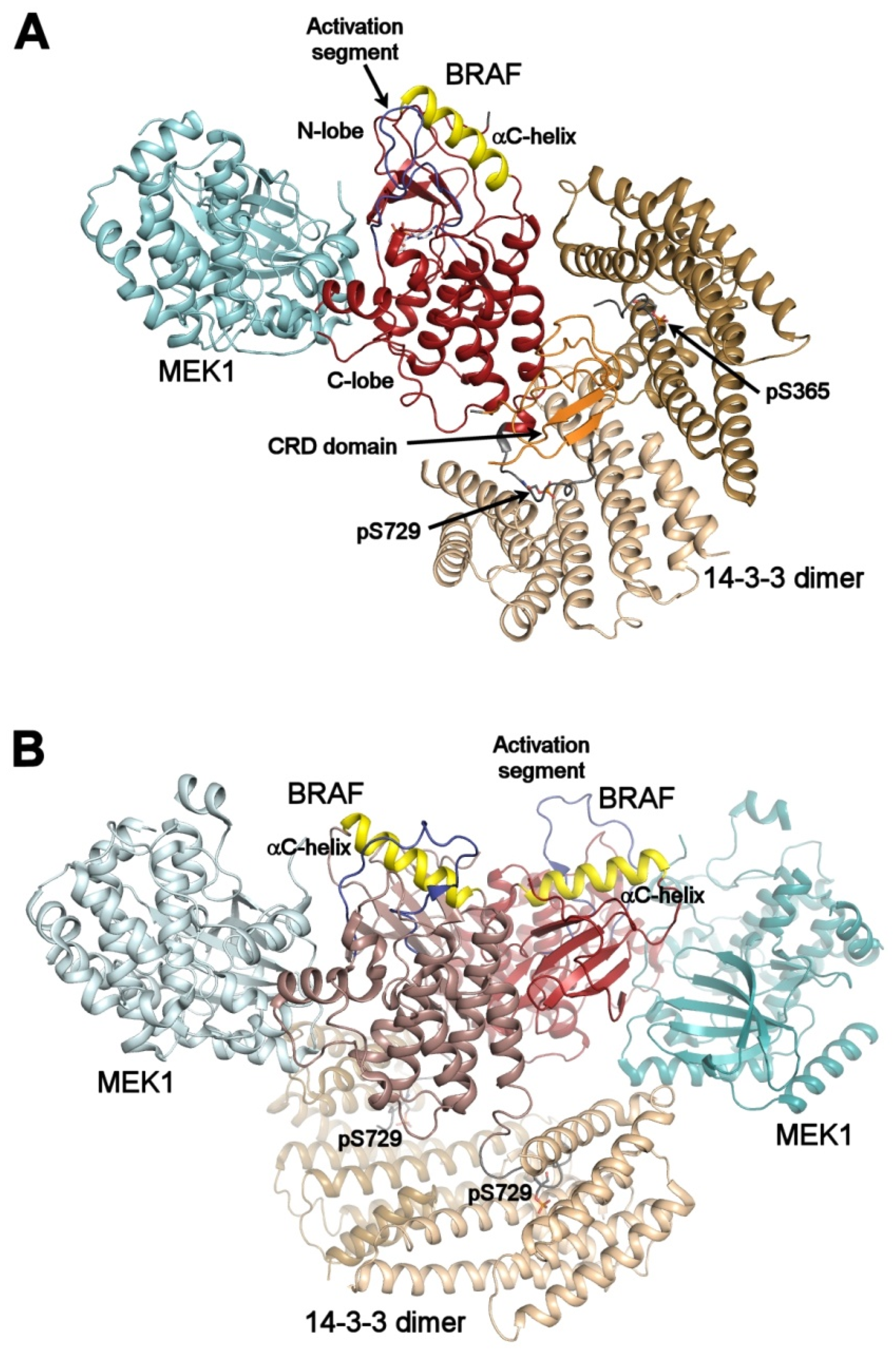
Dimerization Induced by C-Terminal 14–3–3 Binding Is Sufficient for BRAF Kinase Activation | Biochemistry
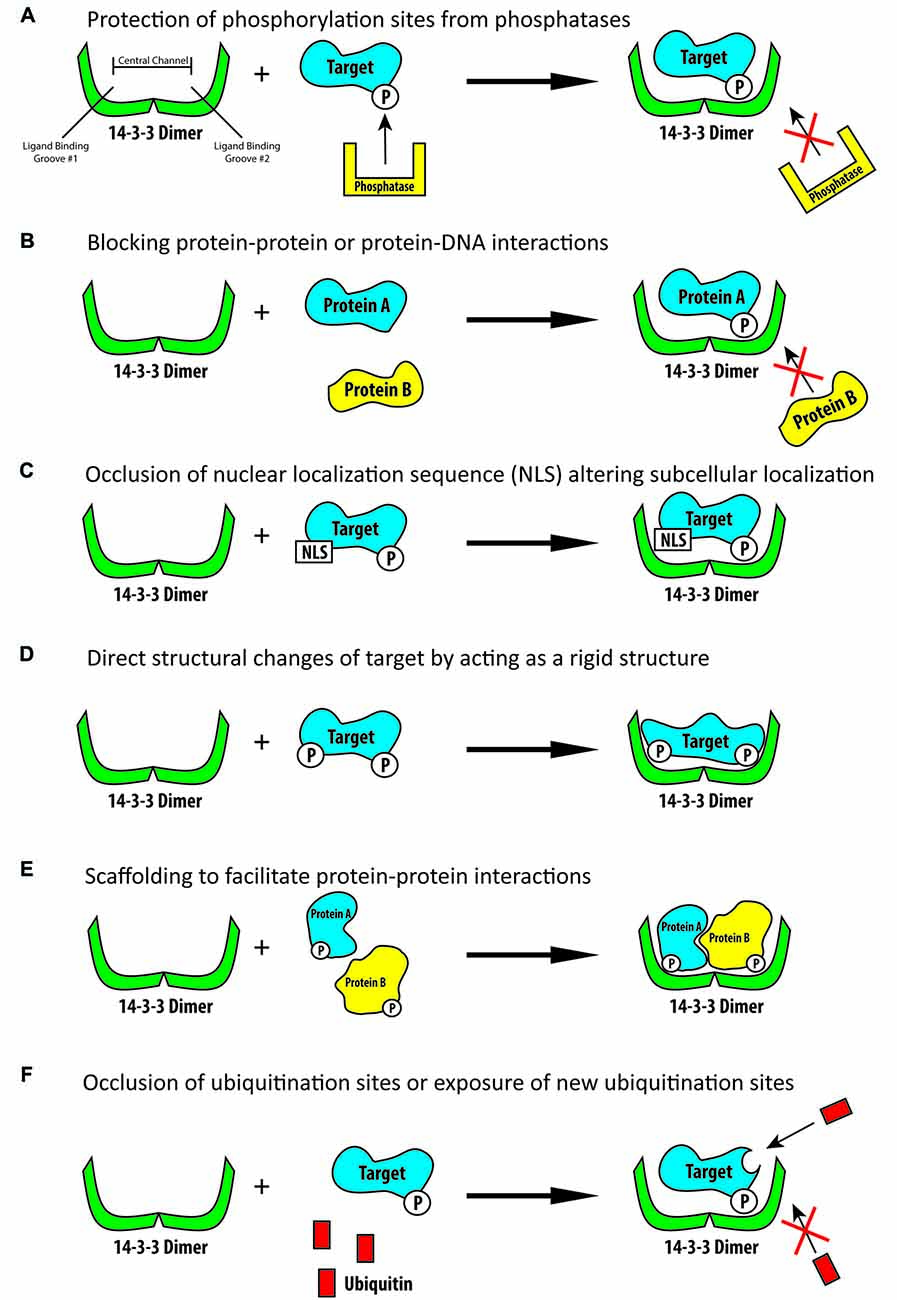
Frontiers | 14-3-3 Proteins in Brain Development: Neurogenesis, Neuronal Migration and Neuromorphogenesis
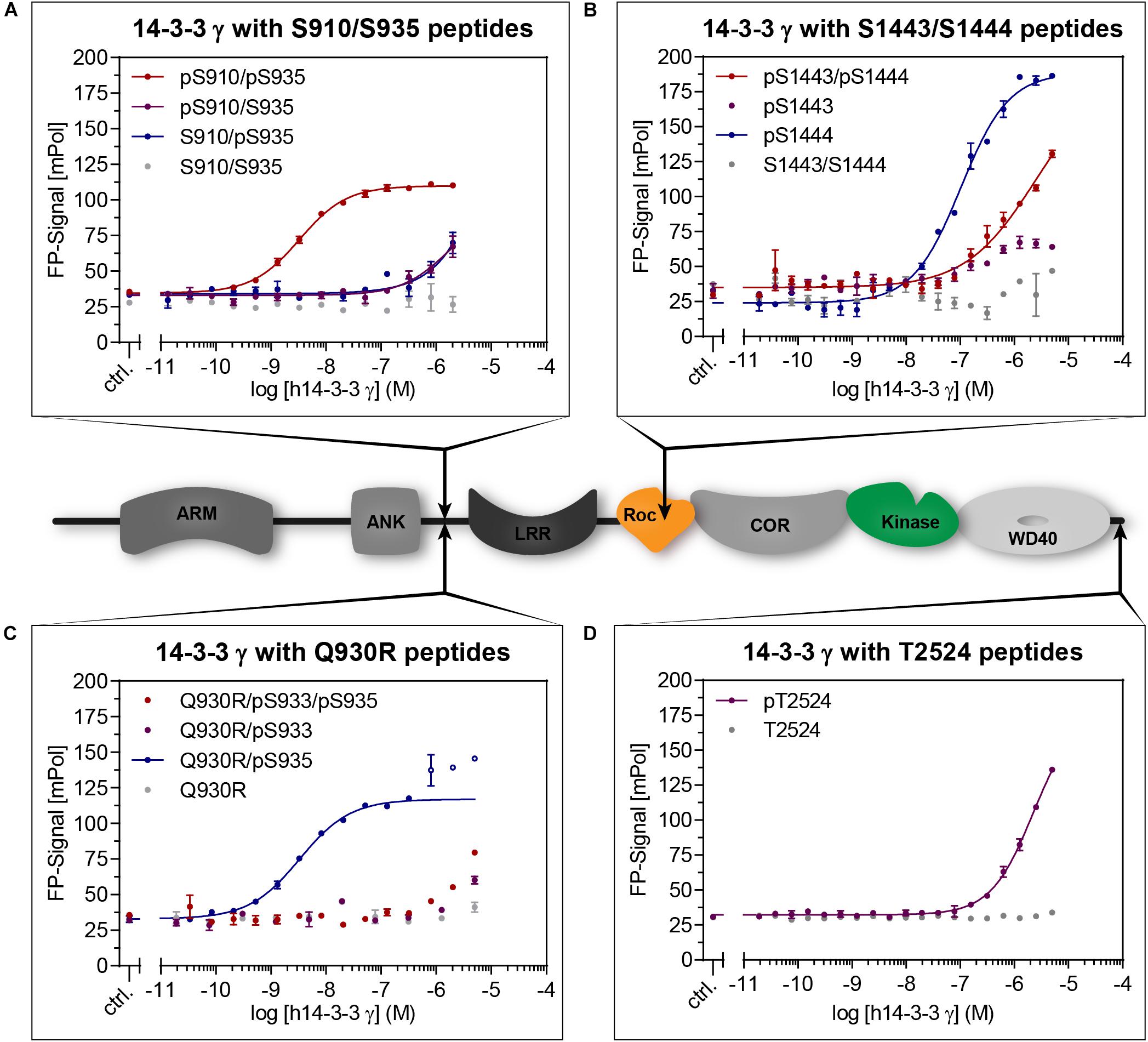
Frontiers | Binding of the Human 14-3-3 Isoforms to Distinct Sites in the Leucine-Rich Repeat Kinase 2

Internal and C-terminal 14-3-3 binding motifs. (A) The internal mode I... | Download Scientific Diagram
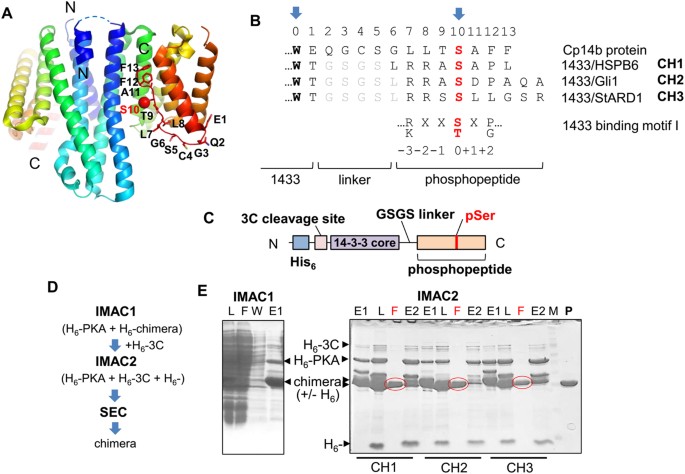
Chimeric 14-3-3 proteins for unraveling interactions with intrinsically disordered partners | Scientific Reports

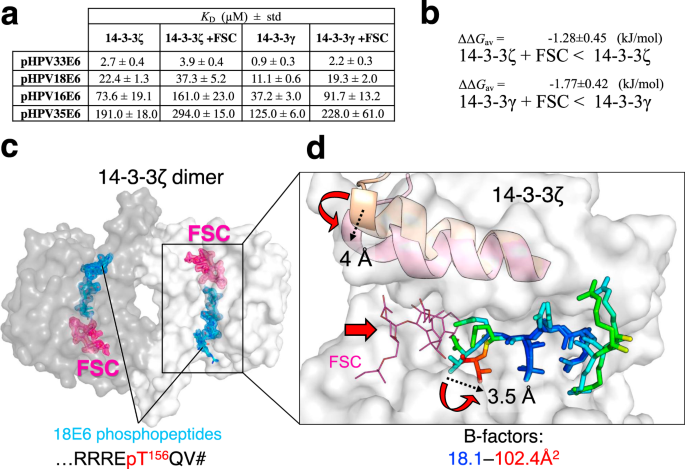
Recognition of high-risk HPV E6 oncoproteins by 14-3-3 proteins studied by interactomics and crystallography | bioRxiv